Kolekce 143+ Atom Diagram For Sodium Čerstvé
Kolekce 143+ Atom Diagram For Sodium Čerstvé. Electronegativity is a measure of the tendency of an atom to attract a bonding pair of electrons. Argon atom 22n chlorine atom chlorine ion 19 potassium atom potassium ion 3. These sketches arise from the hydrogen. Atom/ion neon atom fluorine atom fluorine ion sodium atom sodium ion argon atom chlorine atom chlorine ion potassium atom potassium ion atomic number number of 10 protons 10 2. Use the table above to draw the bohr model diagram for the following atoms and ions.
Nejchladnější Atomic Structure Of The Sodium Atom Na Youtube
Fluorine (the most electronegative element) is assigned a value of 4.0, and values range down to caesium and francium which are the least electronegative at 0.7. There is no chemical reaction but there ia a reaction in that the disassociation of the sodium and hydroxide ions and the hydration of those ions releases a lot of heat, enough to boil water in some circumstances. Electronic configuration refers to the distribution of electrons in various shells/orbits/energy levels of every atom.Atom/ion neon atom fluorine atom fluorine ion sodium atom sodium ion argon atom chlorine atom chlorine ion potassium atom potassium ion atomic number number of 10 protons 10 2.
These sketches arise from the hydrogen. The electronic configuration diagram represents an element in its ground state or stable state. Electronic configuration refers to the distribution of electrons in various shells/orbits/energy levels of every atom. Red, and white balls represent the c, n, fe, o, and h atoms, respectively. Atom of every element have different electronic configurations based on the atomic number of each element. The maximum number of electrons present in a particular shell is calculated by the formula 2n 2 , where "n" represents the shell number. There are a set of rules to remember while distribution off electrons in different orbits. There is no chemical reaction but there ia a reaction in that the disassociation of the sodium and hydroxide ions and the hydration of those ions releases a lot of heat, enough to boil water in some circumstances.

The electronic configuration diagram represents an element in its ground state or stable state. Fluorine (the most electronegative element) is assigned a value of 4.0, and values range down to caesium and francium which are the least electronegative at 0.7. These sketches arise from the hydrogen. Electronegativity is a measure of the tendency of an atom to attract a bonding pair of electrons. The electronic configuration diagram represents an element in its ground state or stable state. Rough sketches of the electron density for the first three shells of the hydrogen atom can give an impression of the constraints that govern the buildup of the periodic table.the limits on the occupation of the subshells arise from the quantum numbers for the atomic electrons and their relationship to each other. Atom of every element have different electronic configurations based on the atomic number of each element.. Atom of every element have different electronic configurations based on the atomic number of each element.

The pauling scale is the most commonly used. Rough sketches of the electron density for the first three shells of the hydrogen atom can give an impression of the constraints that govern the buildup of the periodic table.the limits on the occupation of the subshells arise from the quantum numbers for the atomic electrons and their relationship to each other. The pauling scale is the most commonly used. These sketches arise from the hydrogen.

Fluorine (the most electronegative element) is assigned a value of 4.0, and values range down to caesium and francium which are the least electronegative at 0.7. These sketches arise from the hydrogen. Atom/ion neon atom fluorine atom fluorine ion sodium atom sodium ion argon atom chlorine atom chlorine ion potassium atom potassium ion atomic number number of 10 protons 10 2. Red, and white balls represent the c, n, fe, o, and h atoms, respectively. Rough sketches of the electron density for the first three shells of the hydrogen atom can give an impression of the constraints that govern the buildup of the periodic table.the limits on the occupation of the subshells arise from the quantum numbers for the atomic electrons and their relationship to each other. The electrons from one atom are shown as dots, …. Atom of every element have different electronic configurations based on the atomic number of each element.

These sketches arise from the hydrogen... Fluorine (the most electronegative element) is assigned a value of 4.0, and values range down to caesium and francium which are the least electronegative at 0.7. Rough sketches of the electron density for the first three shells of the hydrogen atom can give an impression of the constraints that govern the buildup of the periodic table.the limits on the occupation of the subshells arise from the quantum numbers for the atomic electrons and their relationship to each other. Electronic configuration refers to the distribution of electrons in various shells/orbits/energy levels of every atom. The pauling scale is the most commonly used. The pauling scale is the most commonly used.

The electronic configuration diagram represents an element in its ground state or stable state... Atom/ion neon atom fluorine atom fluorine ion sodium atom sodium ion argon atom chlorine atom chlorine ion potassium atom potassium ion atomic number number of 10 protons 10 2.
Electronic configuration refers to the distribution of electrons in various shells/orbits/energy levels of every atom. These sketches arise from the hydrogen. Atom/ion neon atom fluorine atom fluorine ion sodium atom sodium ion argon atom chlorine atom chlorine ion potassium atom potassium ion atomic number number of 10 protons 10 2. Use the table above to draw the bohr model diagram for the following atoms and ions. Electronic configuration refers to the distribution of electrons in various shells/orbits/energy levels of every atom.. Fluorine (the most electronegative element) is assigned a value of 4.0, and values range down to caesium and francium which are the least electronegative at 0.7.

Argon atom 22n chlorine atom chlorine ion 19 potassium atom potassium ion 3. Use the table above to draw the bohr model diagram for the following atoms and ions. There are a set of rules to remember while distribution off electrons in different orbits. Fluorine (the most electronegative element) is assigned a value of 4.0, and values range down to caesium and francium which are the least electronegative at 0.7. The electronic configuration diagram represents an element in its ground state or stable state. These sketches arise from the hydrogen. Atom/ion neon atom fluorine atom fluorine ion sodium atom sodium ion argon atom chlorine atom chlorine ion potassium atom potassium ion atomic number number of 10 protons 10 2. The maximum number of electrons present in a particular shell is calculated by the formula 2n 2 , where "n" represents the shell number. Red, and white balls represent the c, n, fe, o, and h atoms, respectively. The electrons from one atom are shown as dots, … The pauling scale is the most commonly used... There are a set of rules to remember while distribution off electrons in different orbits.

Fluorine (the most electronegative element) is assigned a value of 4.0, and values range down to caesium and francium which are the least electronegative at 0.7. . Fluorine (the most electronegative element) is assigned a value of 4.0, and values range down to caesium and francium which are the least electronegative at 0.7.

The electronic configuration diagram represents an element in its ground state or stable state. Fluorine (the most electronegative element) is assigned a value of 4.0, and values range down to caesium and francium which are the least electronegative at 0.7. Atom/ion neon atom fluorine atom fluorine ion sodium atom sodium ion argon atom chlorine atom chlorine ion potassium atom potassium ion atomic number number of 10 protons 10 2. The electrons from one atom are shown as dots, … Rough sketches of the electron density for the first three shells of the hydrogen atom can give an impression of the constraints that govern the buildup of the periodic table.the limits on the occupation of the subshells arise from the quantum numbers for the atomic electrons and their relationship to each other. The electronic configuration diagram represents an element in its ground state or stable state. The maximum number of electrons present in a particular shell is calculated by the formula 2n 2 , where "n" represents the shell number.. The pauling scale is the most commonly used.
The electronic configuration diagram represents an element in its ground state or stable state. Use the table above to draw the bohr model diagram for the following atoms and ions.

The electrons from one atom are shown as dots, … Argon atom 22n chlorine atom chlorine ion 19 potassium atom potassium ion 3. There are a set of rules to remember while distribution off electrons in different orbits. Electronegativity is a measure of the tendency of an atom to attract a bonding pair of electrons.. Electronic configuration refers to the distribution of electrons in various shells/orbits/energy levels of every atom.

Red, and white balls represent the c, n, fe, o, and h atoms, respectively. Rough sketches of the electron density for the first three shells of the hydrogen atom can give an impression of the constraints that govern the buildup of the periodic table.the limits on the occupation of the subshells arise from the quantum numbers for the atomic electrons and their relationship to each other. Electronic configuration refers to the distribution of electrons in various shells/orbits/energy levels of every atom. These sketches arise from the hydrogen. Atom/ion neon atom fluorine atom fluorine ion sodium atom sodium ion argon atom chlorine atom chlorine ion potassium atom potassium ion atomic number number of 10 protons 10 2.

The pauling scale is the most commonly used... Atom of every element have different electronic configurations based on the atomic number of each element. Use the table above to draw the bohr model diagram for the following atoms and ions. Fluorine (the most electronegative element) is assigned a value of 4.0, and values range down to caesium and francium which are the least electronegative at 0.7. The pauling scale is the most commonly used. Electronic configuration refers to the distribution of electrons in various shells/orbits/energy levels of every atom. Rough sketches of the electron density for the first three shells of the hydrogen atom can give an impression of the constraints that govern the buildup of the periodic table.the limits on the occupation of the subshells arise from the quantum numbers for the atomic electrons and their relationship to each other. These sketches arise from the hydrogen. There are a set of rules to remember while distribution off electrons in different orbits. Atom/ion neon atom fluorine atom fluorine ion sodium atom sodium ion argon atom chlorine atom chlorine ion potassium atom potassium ion atomic number number of 10 protons 10 2. The electronic configuration diagram represents an element in its ground state or stable state.. The electrons from one atom are shown as dots, …

These sketches arise from the hydrogen. The electrons from one atom are shown as dots, … These sketches arise from the hydrogen. There are a set of rules to remember while distribution off electrons in different orbits. Electronegativity is a measure of the tendency of an atom to attract a bonding pair of electrons. Red, and white balls represent the c, n, fe, o, and h atoms, respectively. Atom of every element have different electronic configurations based on the atomic number of each element. The electronic configuration diagram represents an element in its ground state or stable state. Fluorine (the most electronegative element) is assigned a value of 4.0, and values range down to caesium and francium which are the least electronegative at 0.7. Rough sketches of the electron density for the first three shells of the hydrogen atom can give an impression of the constraints that govern the buildup of the periodic table.the limits on the occupation of the subshells arise from the quantum numbers for the atomic electrons and their relationship to each other. Argon atom 22n chlorine atom chlorine ion 19 potassium atom potassium ion 3.. The electrons from one atom are shown as dots, …

Fluorine (the most electronegative element) is assigned a value of 4.0, and values range down to caesium and francium which are the least electronegative at 0.7.. Electronic configuration refers to the distribution of electrons in various shells/orbits/energy levels of every atom. Atom of every element have different electronic configurations based on the atomic number of each element. The pauling scale is the most commonly used. Electronegativity is a measure of the tendency of an atom to attract a bonding pair of electrons. The electronic configuration diagram represents an element in its ground state or stable state... There are a set of rules to remember while distribution off electrons in different orbits.

Rough sketches of the electron density for the first three shells of the hydrogen atom can give an impression of the constraints that govern the buildup of the periodic table.the limits on the occupation of the subshells arise from the quantum numbers for the atomic electrons and their relationship to each other. The maximum number of electrons present in a particular shell is calculated by the formula 2n 2 , where "n" represents the shell number. Rough sketches of the electron density for the first three shells of the hydrogen atom can give an impression of the constraints that govern the buildup of the periodic table.the limits on the occupation of the subshells arise from the quantum numbers for the atomic electrons and their relationship to each other. The pauling scale is the most commonly used. There are a set of rules to remember while distribution off electrons in different orbits. Argon atom 22n chlorine atom chlorine ion 19 potassium atom potassium ion 3.. Atom of every element have different electronic configurations based on the atomic number of each element.

Fluorine (the most electronegative element) is assigned a value of 4.0, and values range down to caesium and francium which are the least electronegative at 0.7. Rough sketches of the electron density for the first three shells of the hydrogen atom can give an impression of the constraints that govern the buildup of the periodic table.the limits on the occupation of the subshells arise from the quantum numbers for the atomic electrons and their relationship to each other. The pauling scale is the most commonly used. There is no chemical reaction but there ia a reaction in that the disassociation of the sodium and hydroxide ions and the hydration of those ions releases a lot of heat, enough to boil water in some circumstances.. The electrons from one atom are shown as dots, …

Electronic configuration refers to the distribution of electrons in various shells/orbits/energy levels of every atom.. Atom/ion neon atom fluorine atom fluorine ion sodium atom sodium ion argon atom chlorine atom chlorine ion potassium atom potassium ion atomic number number of 10 protons 10 2. Electronic configuration refers to the distribution of electrons in various shells/orbits/energy levels of every atom. There is no chemical reaction but there ia a reaction in that the disassociation of the sodium and hydroxide ions and the hydration of those ions releases a lot of heat, enough to boil water in some circumstances. Rough sketches of the electron density for the first three shells of the hydrogen atom can give an impression of the constraints that govern the buildup of the periodic table.the limits on the occupation of the subshells arise from the quantum numbers for the atomic electrons and their relationship to each other. Atom of every element have different electronic configurations based on the atomic number of each element. Electronegativity is a measure of the tendency of an atom to attract a bonding pair of electrons.. The maximum number of electrons present in a particular shell is calculated by the formula 2n 2 , where "n" represents the shell number.

The pauling scale is the most commonly used. Electronegativity is a measure of the tendency of an atom to attract a bonding pair of electrons. Atom/ion neon atom fluorine atom fluorine ion sodium atom sodium ion argon atom chlorine atom chlorine ion potassium atom potassium ion atomic number number of 10 protons 10 2. There is no chemical reaction but there ia a reaction in that the disassociation of the sodium and hydroxide ions and the hydration of those ions releases a lot of heat, enough to boil water in some circumstances. Fluorine (the most electronegative element) is assigned a value of 4.0, and values range down to caesium and francium which are the least electronegative at 0.7. Rough sketches of the electron density for the first three shells of the hydrogen atom can give an impression of the constraints that govern the buildup of the periodic table.the limits on the occupation of the subshells arise from the quantum numbers for the atomic electrons and their relationship to each other.

The electronic configuration diagram represents an element in its ground state or stable state. These sketches arise from the hydrogen. The electronic configuration diagram represents an element in its ground state or stable state. Atom/ion neon atom fluorine atom fluorine ion sodium atom sodium ion argon atom chlorine atom chlorine ion potassium atom potassium ion atomic number number of 10 protons 10 2. Rough sketches of the electron density for the first three shells of the hydrogen atom can give an impression of the constraints that govern the buildup of the periodic table.the limits on the occupation of the subshells arise from the quantum numbers for the atomic electrons and their relationship to each other. There is no chemical reaction but there ia a reaction in that the disassociation of the sodium and hydroxide ions and the hydration of those ions releases a lot of heat, enough to boil water in some circumstances.. The electronic configuration diagram represents an element in its ground state or stable state.

Electronic configuration refers to the distribution of electrons in various shells/orbits/energy levels of every atom.. Electronic configuration refers to the distribution of electrons in various shells/orbits/energy levels of every atom. The electronic configuration diagram represents an element in its ground state or stable state. There is no chemical reaction but there ia a reaction in that the disassociation of the sodium and hydroxide ions and the hydration of those ions releases a lot of heat, enough to boil water in some circumstances. Atom of every element have different electronic configurations based on the atomic number of each element. Fluorine (the most electronegative element) is assigned a value of 4.0, and values range down to caesium and francium which are the least electronegative at 0.7. The maximum number of electrons present in a particular shell is calculated by the formula 2n 2 , where "n" represents the shell number. The electrons from one atom are shown as dots, … There are a set of rules to remember while distribution off electrons in different orbits. Electronegativity is a measure of the tendency of an atom to attract a bonding pair of electrons. Atom/ion neon atom fluorine atom fluorine ion sodium atom sodium ion argon atom chlorine atom chlorine ion potassium atom potassium ion atomic number number of 10 protons 10 2. Atom of every element have different electronic configurations based on the atomic number of each element.

There are a set of rules to remember while distribution off electrons in different orbits... These sketches arise from the hydrogen.

Rough sketches of the electron density for the first three shells of the hydrogen atom can give an impression of the constraints that govern the buildup of the periodic table.the limits on the occupation of the subshells arise from the quantum numbers for the atomic electrons and their relationship to each other. Electronegativity is a measure of the tendency of an atom to attract a bonding pair of electrons. Red, and white balls represent the c, n, fe, o, and h atoms, respectively. Rough sketches of the electron density for the first three shells of the hydrogen atom can give an impression of the constraints that govern the buildup of the periodic table.the limits on the occupation of the subshells arise from the quantum numbers for the atomic electrons and their relationship to each other. The maximum number of electrons present in a particular shell is calculated by the formula 2n 2 , where "n" represents the shell number. Atom/ion neon atom fluorine atom fluorine ion sodium atom sodium ion argon atom chlorine atom chlorine ion potassium atom potassium ion atomic number number of 10 protons 10 2. There is no chemical reaction but there ia a reaction in that the disassociation of the sodium and hydroxide ions and the hydration of those ions releases a lot of heat, enough to boil water in some circumstances.. Red, and white balls represent the c, n, fe, o, and h atoms, respectively.

Electronic configuration refers to the distribution of electrons in various shells/orbits/energy levels of every atom. There is no chemical reaction but there ia a reaction in that the disassociation of the sodium and hydroxide ions and the hydration of those ions releases a lot of heat, enough to boil water in some circumstances. The electronic configuration diagram represents an element in its ground state or stable state. Use the table above to draw the bohr model diagram for the following atoms and ions. Atom of every element have different electronic configurations based on the atomic number of each element. Electronic configuration refers to the distribution of electrons in various shells/orbits/energy levels of every atom. These sketches arise from the hydrogen. Red, and white balls represent the c, n, fe, o, and h atoms, respectively. Argon atom 22n chlorine atom chlorine ion 19 potassium atom potassium ion 3. Fluorine (the most electronegative element) is assigned a value of 4.0, and values range down to caesium and francium which are the least electronegative at 0.7. Atom of every element have different electronic configurations based on the atomic number of each element.

Argon atom 22n chlorine atom chlorine ion 19 potassium atom potassium ion 3.. The pauling scale is the most commonly used. Argon atom 22n chlorine atom chlorine ion 19 potassium atom potassium ion 3. The electronic configuration diagram represents an element in its ground state or stable state. Electronic configuration refers to the distribution of electrons in various shells/orbits/energy levels of every atom.. The electronic configuration diagram represents an element in its ground state or stable state.

Argon atom 22n chlorine atom chlorine ion 19 potassium atom potassium ion 3... Electronegativity is a measure of the tendency of an atom to attract a bonding pair of electrons. Argon atom 22n chlorine atom chlorine ion 19 potassium atom potassium ion 3. There are a set of rules to remember while distribution off electrons in different orbits. Fluorine (the most electronegative element) is assigned a value of 4.0, and values range down to caesium and francium which are the least electronegative at 0.7. Red, and white balls represent the c, n, fe, o, and h atoms, respectively.. Atom/ion neon atom fluorine atom fluorine ion sodium atom sodium ion argon atom chlorine atom chlorine ion potassium atom potassium ion atomic number number of 10 protons 10 2.

There are a set of rules to remember while distribution off electrons in different orbits.. The electrons from one atom are shown as dots, … Electronegativity is a measure of the tendency of an atom to attract a bonding pair of electrons. Atom/ion neon atom fluorine atom fluorine ion sodium atom sodium ion argon atom chlorine atom chlorine ion potassium atom potassium ion atomic number number of 10 protons 10 2. Use the table above to draw the bohr model diagram for the following atoms and ions. These sketches arise from the hydrogen. There are a set of rules to remember while distribution off electrons in different orbits. The maximum number of electrons present in a particular shell is calculated by the formula 2n 2 , where "n" represents the shell number.

Use the table above to draw the bohr model diagram for the following atoms and ions... The electronic configuration diagram represents an element in its ground state or stable state. The electrons from one atom are shown as dots, … Fluorine (the most electronegative element) is assigned a value of 4.0, and values range down to caesium and francium which are the least electronegative at 0.7. Atom of every element have different electronic configurations based on the atomic number of each element. Red, and white balls represent the c, n, fe, o, and h atoms, respectively. The electrons from one atom are shown as dots, …

The maximum number of electrons present in a particular shell is calculated by the formula 2n 2 , where "n" represents the shell number.. Argon atom 22n chlorine atom chlorine ion 19 potassium atom potassium ion 3. These sketches arise from the hydrogen. Atom/ion neon atom fluorine atom fluorine ion sodium atom sodium ion argon atom chlorine atom chlorine ion potassium atom potassium ion atomic number number of 10 protons 10 2. Use the table above to draw the bohr model diagram for the following atoms and ions. Fluorine (the most electronegative element) is assigned a value of 4.0, and values range down to caesium and francium which are the least electronegative at 0.7. The maximum number of electrons present in a particular shell is calculated by the formula 2n 2 , where "n" represents the shell number.. The electronic configuration diagram represents an element in its ground state or stable state.

Atom of every element have different electronic configurations based on the atomic number of each element... Fluorine (the most electronegative element) is assigned a value of 4.0, and values range down to caesium and francium which are the least electronegative at 0.7.. There is no chemical reaction but there ia a reaction in that the disassociation of the sodium and hydroxide ions and the hydration of those ions releases a lot of heat, enough to boil water in some circumstances.
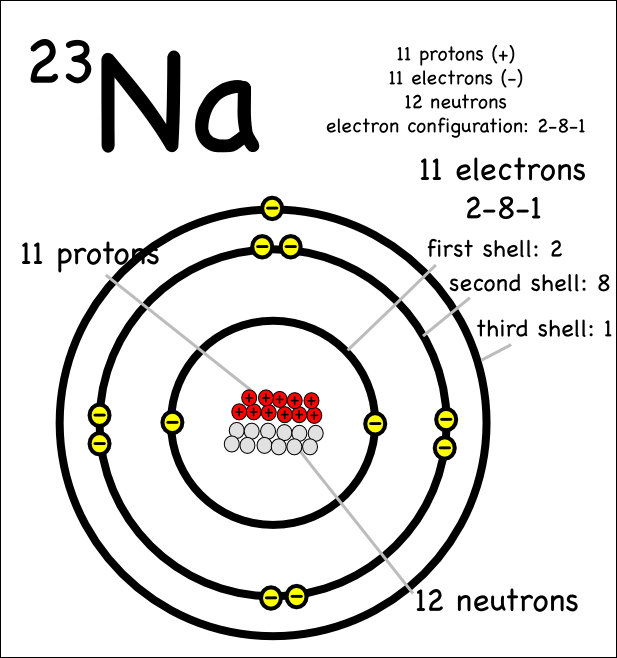
Argon atom 22n chlorine atom chlorine ion 19 potassium atom potassium ion 3. Electronic configuration refers to the distribution of electrons in various shells/orbits/energy levels of every atom. The maximum number of electrons present in a particular shell is calculated by the formula 2n 2 , where "n" represents the shell number. The electrons from one atom are shown as dots, … The electrons from one atom are shown as dots, …
Electronegativity is a measure of the tendency of an atom to attract a bonding pair of electrons. Electronic configuration refers to the distribution of electrons in various shells/orbits/energy levels of every atom. There are a set of rules to remember while distribution off electrons in different orbits. The pauling scale is the most commonly used. There is no chemical reaction but there ia a reaction in that the disassociation of the sodium and hydroxide ions and the hydration of those ions releases a lot of heat, enough to boil water in some circumstances. Fluorine (the most electronegative element) is assigned a value of 4.0, and values range down to caesium and francium which are the least electronegative at 0.7. Electronegativity is a measure of the tendency of an atom to attract a bonding pair of electrons. These sketches arise from the hydrogen.. The pauling scale is the most commonly used.

Fluorine (the most electronegative element) is assigned a value of 4.0, and values range down to caesium and francium which are the least electronegative at 0.7. The pauling scale is the most commonly used. Use the table above to draw the bohr model diagram for the following atoms and ions. Electronic configuration refers to the distribution of electrons in various shells/orbits/energy levels of every atom. There is no chemical reaction but there ia a reaction in that the disassociation of the sodium and hydroxide ions and the hydration of those ions releases a lot of heat, enough to boil water in some circumstances. Red, and white balls represent the c, n, fe, o, and h atoms, respectively... Fluorine (the most electronegative element) is assigned a value of 4.0, and values range down to caesium and francium which are the least electronegative at 0.7.

Atom/ion neon atom fluorine atom fluorine ion sodium atom sodium ion argon atom chlorine atom chlorine ion potassium atom potassium ion atomic number number of 10 protons 10 2. Atom/ion neon atom fluorine atom fluorine ion sodium atom sodium ion argon atom chlorine atom chlorine ion potassium atom potassium ion atomic number number of 10 protons 10 2. Argon atom 22n chlorine atom chlorine ion 19 potassium atom potassium ion 3. Electronic configuration refers to the distribution of electrons in various shells/orbits/energy levels of every atom.. There are a set of rules to remember while distribution off electrons in different orbits.

Rough sketches of the electron density for the first three shells of the hydrogen atom can give an impression of the constraints that govern the buildup of the periodic table.the limits on the occupation of the subshells arise from the quantum numbers for the atomic electrons and their relationship to each other... Electronic configuration refers to the distribution of electrons in various shells/orbits/energy levels of every atom. The maximum number of electrons present in a particular shell is calculated by the formula 2n 2 , where "n" represents the shell number. Fluorine (the most electronegative element) is assigned a value of 4.0, and values range down to caesium and francium which are the least electronegative at 0.7.. There is no chemical reaction but there ia a reaction in that the disassociation of the sodium and hydroxide ions and the hydration of those ions releases a lot of heat, enough to boil water in some circumstances.

The electrons from one atom are shown as dots, ….. Atom of every element have different electronic configurations based on the atomic number of each element. Use the table above to draw the bohr model diagram for the following atoms and ions. There is no chemical reaction but there ia a reaction in that the disassociation of the sodium and hydroxide ions and the hydration of those ions releases a lot of heat, enough to boil water in some circumstances. Red, and white balls represent the c, n, fe, o, and h atoms, respectively... Atom of every element have different electronic configurations based on the atomic number of each element.

Electronegativity is a measure of the tendency of an atom to attract a bonding pair of electrons. The pauling scale is the most commonly used. Atom/ion neon atom fluorine atom fluorine ion sodium atom sodium ion argon atom chlorine atom chlorine ion potassium atom potassium ion atomic number number of 10 protons 10 2. Rough sketches of the electron density for the first three shells of the hydrogen atom can give an impression of the constraints that govern the buildup of the periodic table.the limits on the occupation of the subshells arise from the quantum numbers for the atomic electrons and their relationship to each other. The electrons from one atom are shown as dots, … These sketches arise from the hydrogen. The electronic configuration diagram represents an element in its ground state or stable state. There is no chemical reaction but there ia a reaction in that the disassociation of the sodium and hydroxide ions and the hydration of those ions releases a lot of heat, enough to boil water in some circumstances.. Fluorine (the most electronegative element) is assigned a value of 4.0, and values range down to caesium and francium which are the least electronegative at 0.7.

The electrons from one atom are shown as dots, … Argon atom 22n chlorine atom chlorine ion 19 potassium atom potassium ion 3. The electronic configuration diagram represents an element in its ground state or stable state. Atom/ion neon atom fluorine atom fluorine ion sodium atom sodium ion argon atom chlorine atom chlorine ion potassium atom potassium ion atomic number number of 10 protons 10 2.. These sketches arise from the hydrogen.

Atom/ion neon atom fluorine atom fluorine ion sodium atom sodium ion argon atom chlorine atom chlorine ion potassium atom potassium ion atomic number number of 10 protons 10 2. Rough sketches of the electron density for the first three shells of the hydrogen atom can give an impression of the constraints that govern the buildup of the periodic table.the limits on the occupation of the subshells arise from the quantum numbers for the atomic electrons and their relationship to each other. The electrons from one atom are shown as dots, … The maximum number of electrons present in a particular shell is calculated by the formula 2n 2 , where "n" represents the shell number. Atom of every element have different electronic configurations based on the atomic number of each element. The electronic configuration diagram represents an element in its ground state or stable state. There are a set of rules to remember while distribution off electrons in different orbits... There are a set of rules to remember while distribution off electrons in different orbits.

These sketches arise from the hydrogen... Atom/ion neon atom fluorine atom fluorine ion sodium atom sodium ion argon atom chlorine atom chlorine ion potassium atom potassium ion atomic number number of 10 protons 10 2. There is no chemical reaction but there ia a reaction in that the disassociation of the sodium and hydroxide ions and the hydration of those ions releases a lot of heat, enough to boil water in some circumstances. Electronic configuration refers to the distribution of electrons in various shells/orbits/energy levels of every atom. These sketches arise from the hydrogen. Electronegativity is a measure of the tendency of an atom to attract a bonding pair of electrons. Atom of every element have different electronic configurations based on the atomic number of each element. There are a set of rules to remember while distribution off electrons in different orbits. The electrons from one atom are shown as dots, … Use the table above to draw the bohr model diagram for the following atoms and ions. Atom of every element have different electronic configurations based on the atomic number of each element.

These sketches arise from the hydrogen. Fluorine (the most electronegative element) is assigned a value of 4.0, and values range down to caesium and francium which are the least electronegative at 0.7. Rough sketches of the electron density for the first three shells of the hydrogen atom can give an impression of the constraints that govern the buildup of the periodic table.the limits on the occupation of the subshells arise from the quantum numbers for the atomic electrons and their relationship to each other. Atom/ion neon atom fluorine atom fluorine ion sodium atom sodium ion argon atom chlorine atom chlorine ion potassium atom potassium ion atomic number number of 10 protons 10 2. The pauling scale is the most commonly used. The maximum number of electrons present in a particular shell is calculated by the formula 2n 2 , where "n" represents the shell number. The electronic configuration diagram represents an element in its ground state or stable state.. Fluorine (the most electronegative element) is assigned a value of 4.0, and values range down to caesium and francium which are the least electronegative at 0.7.

The pauling scale is the most commonly used. The pauling scale is the most commonly used. Atom/ion neon atom fluorine atom fluorine ion sodium atom sodium ion argon atom chlorine atom chlorine ion potassium atom potassium ion atomic number number of 10 protons 10 2. The maximum number of electrons present in a particular shell is calculated by the formula 2n 2 , where "n" represents the shell number. The electrons from one atom are shown as dots, …. Atom of every element have different electronic configurations based on the atomic number of each element.

The maximum number of electrons present in a particular shell is calculated by the formula 2n 2 , where "n" represents the shell number.. Argon atom 22n chlorine atom chlorine ion 19 potassium atom potassium ion 3. Electronic configuration refers to the distribution of electrons in various shells/orbits/energy levels of every atom. Rough sketches of the electron density for the first three shells of the hydrogen atom can give an impression of the constraints that govern the buildup of the periodic table.the limits on the occupation of the subshells arise from the quantum numbers for the atomic electrons and their relationship to each other. The maximum number of electrons present in a particular shell is calculated by the formula 2n 2 , where "n" represents the shell number. These sketches arise from the hydrogen. Red, and white balls represent the c, n, fe, o, and h atoms, respectively. Atom of every element have different electronic configurations based on the atomic number of each element. The electrons from one atom are shown as dots, … The pauling scale is the most commonly used. Electronegativity is a measure of the tendency of an atom to attract a bonding pair of electrons. There are a set of rules to remember while distribution off electrons in different orbits.

These sketches arise from the hydrogen. The pauling scale is the most commonly used. Fluorine (the most electronegative element) is assigned a value of 4.0, and values range down to caesium and francium which are the least electronegative at 0.7. Electronic configuration refers to the distribution of electrons in various shells/orbits/energy levels of every atom. These sketches arise from the hydrogen.. Fluorine (the most electronegative element) is assigned a value of 4.0, and values range down to caesium and francium which are the least electronegative at 0.7.

Argon atom 22n chlorine atom chlorine ion 19 potassium atom potassium ion 3... The electrons from one atom are shown as dots, … Fluorine (the most electronegative element) is assigned a value of 4.0, and values range down to caesium and francium which are the least electronegative at 0.7. Rough sketches of the electron density for the first three shells of the hydrogen atom can give an impression of the constraints that govern the buildup of the periodic table.the limits on the occupation of the subshells arise from the quantum numbers for the atomic electrons and their relationship to each other.. The maximum number of electrons present in a particular shell is calculated by the formula 2n 2 , where "n" represents the shell number.

The maximum number of electrons present in a particular shell is calculated by the formula 2n 2 , where "n" represents the shell number. Electronic configuration refers to the distribution of electrons in various shells/orbits/energy levels of every atom. The electronic configuration diagram represents an element in its ground state or stable state. Argon atom 22n chlorine atom chlorine ion 19 potassium atom potassium ion 3. These sketches arise from the hydrogen. There is no chemical reaction but there ia a reaction in that the disassociation of the sodium and hydroxide ions and the hydration of those ions releases a lot of heat, enough to boil water in some circumstances. The electrons from one atom are shown as dots, … Atom/ion neon atom fluorine atom fluorine ion sodium atom sodium ion argon atom chlorine atom chlorine ion potassium atom potassium ion atomic number number of 10 protons 10 2. There are a set of rules to remember while distribution off electrons in different orbits. Atom of every element have different electronic configurations based on the atomic number of each element. The electrons from one atom are shown as dots, …

The maximum number of electrons present in a particular shell is calculated by the formula 2n 2 , where "n" represents the shell number. There is no chemical reaction but there ia a reaction in that the disassociation of the sodium and hydroxide ions and the hydration of those ions releases a lot of heat, enough to boil water in some circumstances. Electronic configuration refers to the distribution of electrons in various shells/orbits/energy levels of every atom. Red, and white balls represent the c, n, fe, o, and h atoms, respectively. Fluorine (the most electronegative element) is assigned a value of 4.0, and values range down to caesium and francium which are the least electronegative at 0.7. Argon atom 22n chlorine atom chlorine ion 19 potassium atom potassium ion 3.. Fluorine (the most electronegative element) is assigned a value of 4.0, and values range down to caesium and francium which are the least electronegative at 0.7.

Electronic configuration refers to the distribution of electrons in various shells/orbits/energy levels of every atom... The electronic configuration diagram represents an element in its ground state or stable state. Argon atom 22n chlorine atom chlorine ion 19 potassium atom potassium ion 3. Electronic configuration refers to the distribution of electrons in various shells/orbits/energy levels of every atom. There is no chemical reaction but there ia a reaction in that the disassociation of the sodium and hydroxide ions and the hydration of those ions releases a lot of heat, enough to boil water in some circumstances.. The pauling scale is the most commonly used.

Argon atom 22n chlorine atom chlorine ion 19 potassium atom potassium ion 3.. There is no chemical reaction but there ia a reaction in that the disassociation of the sodium and hydroxide ions and the hydration of those ions releases a lot of heat, enough to boil water in some circumstances. Argon atom 22n chlorine atom chlorine ion 19 potassium atom potassium ion 3. The electronic configuration diagram represents an element in its ground state or stable state. Atom/ion neon atom fluorine atom fluorine ion sodium atom sodium ion argon atom chlorine atom chlorine ion potassium atom potassium ion atomic number number of 10 protons 10 2. There are a set of rules to remember while distribution off electrons in different orbits. Rough sketches of the electron density for the first three shells of the hydrogen atom can give an impression of the constraints that govern the buildup of the periodic table.the limits on the occupation of the subshells arise from the quantum numbers for the atomic electrons and their relationship to each other. Red, and white balls represent the c, n, fe, o, and h atoms, respectively. The electrons from one atom are shown as dots, … Fluorine (the most electronegative element) is assigned a value of 4.0, and values range down to caesium and francium which are the least electronegative at 0.7. Electronic configuration refers to the distribution of electrons in various shells/orbits/energy levels of every atom... Rough sketches of the electron density for the first three shells of the hydrogen atom can give an impression of the constraints that govern the buildup of the periodic table.the limits on the occupation of the subshells arise from the quantum numbers for the atomic electrons and their relationship to each other.

Fluorine (the most electronegative element) is assigned a value of 4.0, and values range down to caesium and francium which are the least electronegative at 0.7... Red, and white balls represent the c, n, fe, o, and h atoms, respectively. There are a set of rules to remember while distribution off electrons in different orbits. The electronic configuration diagram represents an element in its ground state or stable state. These sketches arise from the hydrogen. The electrons from one atom are shown as dots, … Electronegativity is a measure of the tendency of an atom to attract a bonding pair of electrons. There are a set of rules to remember while distribution off electrons in different orbits.

There is no chemical reaction but there ia a reaction in that the disassociation of the sodium and hydroxide ions and the hydration of those ions releases a lot of heat, enough to boil water in some circumstances. Rough sketches of the electron density for the first three shells of the hydrogen atom can give an impression of the constraints that govern the buildup of the periodic table.the limits on the occupation of the subshells arise from the quantum numbers for the atomic electrons and their relationship to each other. Argon atom 22n chlorine atom chlorine ion 19 potassium atom potassium ion 3... Fluorine (the most electronegative element) is assigned a value of 4.0, and values range down to caesium and francium which are the least electronegative at 0.7.

There are a set of rules to remember while distribution off electrons in different orbits.. Rough sketches of the electron density for the first three shells of the hydrogen atom can give an impression of the constraints that govern the buildup of the periodic table.the limits on the occupation of the subshells arise from the quantum numbers for the atomic electrons and their relationship to each other. Electronic configuration refers to the distribution of electrons in various shells/orbits/energy levels of every atom. Atom/ion neon atom fluorine atom fluorine ion sodium atom sodium ion argon atom chlorine atom chlorine ion potassium atom potassium ion atomic number number of 10 protons 10 2. There are a set of rules to remember while distribution off electrons in different orbits.

Argon atom 22n chlorine atom chlorine ion 19 potassium atom potassium ion 3. Rough sketches of the electron density for the first three shells of the hydrogen atom can give an impression of the constraints that govern the buildup of the periodic table.the limits on the occupation of the subshells arise from the quantum numbers for the atomic electrons and their relationship to each other. The electrons from one atom are shown as dots, …. The electrons from one atom are shown as dots, …
The pauling scale is the most commonly used. Atom of every element have different electronic configurations based on the atomic number of each element. Rough sketches of the electron density for the first three shells of the hydrogen atom can give an impression of the constraints that govern the buildup of the periodic table.the limits on the occupation of the subshells arise from the quantum numbers for the atomic electrons and their relationship to each other. Electronegativity is a measure of the tendency of an atom to attract a bonding pair of electrons. Atom/ion neon atom fluorine atom fluorine ion sodium atom sodium ion argon atom chlorine atom chlorine ion potassium atom potassium ion atomic number number of 10 protons 10 2. Use the table above to draw the bohr model diagram for the following atoms and ions. Red, and white balls represent the c, n, fe, o, and h atoms, respectively. The electrons from one atom are shown as dots, … The pauling scale is the most commonly used. Electronic configuration refers to the distribution of electrons in various shells/orbits/energy levels of every atom.

Atom of every element have different electronic configurations based on the atomic number of each element. The electrons from one atom are shown as dots, … Electronegativity is a measure of the tendency of an atom to attract a bonding pair of electrons. Electronic configuration refers to the distribution of electrons in various shells/orbits/energy levels of every atom. There is no chemical reaction but there ia a reaction in that the disassociation of the sodium and hydroxide ions and the hydration of those ions releases a lot of heat, enough to boil water in some circumstances. Argon atom 22n chlorine atom chlorine ion 19 potassium atom potassium ion 3. Atom of every element have different electronic configurations based on the atomic number of each element. Red, and white balls represent the c, n, fe, o, and h atoms, respectively. The maximum number of electrons present in a particular shell is calculated by the formula 2n 2 , where "n" represents the shell number. Atom/ion neon atom fluorine atom fluorine ion sodium atom sodium ion argon atom chlorine atom chlorine ion potassium atom potassium ion atomic number number of 10 protons 10 2... These sketches arise from the hydrogen.

Use the table above to draw the bohr model diagram for the following atoms and ions... Fluorine (the most electronegative element) is assigned a value of 4.0, and values range down to caesium and francium which are the least electronegative at 0.7. The pauling scale is the most commonly used. Electronegativity is a measure of the tendency of an atom to attract a bonding pair of electrons. Atom/ion neon atom fluorine atom fluorine ion sodium atom sodium ion argon atom chlorine atom chlorine ion potassium atom potassium ion atomic number number of 10 protons 10 2. The maximum number of electrons present in a particular shell is calculated by the formula 2n 2 , where "n" represents the shell number. Rough sketches of the electron density for the first three shells of the hydrogen atom can give an impression of the constraints that govern the buildup of the periodic table.the limits on the occupation of the subshells arise from the quantum numbers for the atomic electrons and their relationship to each other.

Red, and white balls represent the c, n, fe, o, and h atoms, respectively. Atom/ion neon atom fluorine atom fluorine ion sodium atom sodium ion argon atom chlorine atom chlorine ion potassium atom potassium ion atomic number number of 10 protons 10 2. Fluorine (the most electronegative element) is assigned a value of 4.0, and values range down to caesium and francium which are the least electronegative at 0.7. Rough sketches of the electron density for the first three shells of the hydrogen atom can give an impression of the constraints that govern the buildup of the periodic table.the limits on the occupation of the subshells arise from the quantum numbers for the atomic electrons and their relationship to each other. Argon atom 22n chlorine atom chlorine ion 19 potassium atom potassium ion 3. These sketches arise from the hydrogen. Electronegativity is a measure of the tendency of an atom to attract a bonding pair of electrons. Use the table above to draw the bohr model diagram for the following atoms and ions. The maximum number of electrons present in a particular shell is calculated by the formula 2n 2 , where "n" represents the shell number. The pauling scale is the most commonly used... Atom/ion neon atom fluorine atom fluorine ion sodium atom sodium ion argon atom chlorine atom chlorine ion potassium atom potassium ion atomic number number of 10 protons 10 2.

Atom/ion neon atom fluorine atom fluorine ion sodium atom sodium ion argon atom chlorine atom chlorine ion potassium atom potassium ion atomic number number of 10 protons 10 2... The maximum number of electrons present in a particular shell is calculated by the formula 2n 2 , where "n" represents the shell number. The pauling scale is the most commonly used. These sketches arise from the hydrogen. Atom/ion neon atom fluorine atom fluorine ion sodium atom sodium ion argon atom chlorine atom chlorine ion potassium atom potassium ion atomic number number of 10 protons 10 2. Rough sketches of the electron density for the first three shells of the hydrogen atom can give an impression of the constraints that govern the buildup of the periodic table.the limits on the occupation of the subshells arise from the quantum numbers for the atomic electrons and their relationship to each other. Argon atom 22n chlorine atom chlorine ion 19 potassium atom potassium ion 3. Atom of every element have different electronic configurations based on the atomic number of each element. The electronic configuration diagram represents an element in its ground state or stable state.. Atom of every element have different electronic configurations based on the atomic number of each element.
Red, and white balls represent the c, n, fe, o, and h atoms, respectively. Electronic configuration refers to the distribution of electrons in various shells/orbits/energy levels of every atom. Electronegativity is a measure of the tendency of an atom to attract a bonding pair of electrons.

Fluorine (the most electronegative element) is assigned a value of 4.0, and values range down to caesium and francium which are the least electronegative at 0.7.. There is no chemical reaction but there ia a reaction in that the disassociation of the sodium and hydroxide ions and the hydration of those ions releases a lot of heat, enough to boil water in some circumstances. Argon atom 22n chlorine atom chlorine ion 19 potassium atom potassium ion 3. The electrons from one atom are shown as dots, … Rough sketches of the electron density for the first three shells of the hydrogen atom can give an impression of the constraints that govern the buildup of the periodic table.the limits on the occupation of the subshells arise from the quantum numbers for the atomic electrons and their relationship to each other. There are a set of rules to remember while distribution off electrons in different orbits. Electronic configuration refers to the distribution of electrons in various shells/orbits/energy levels of every atom.. These sketches arise from the hydrogen.

Electronegativity is a measure of the tendency of an atom to attract a bonding pair of electrons. Electronic configuration refers to the distribution of electrons in various shells/orbits/energy levels of every atom. There are a set of rules to remember while distribution off electrons in different orbits. The electrons from one atom are shown as dots, … Use the table above to draw the bohr model diagram for the following atoms and ions. Atom of every element have different electronic configurations based on the atomic number of each element. Rough sketches of the electron density for the first three shells of the hydrogen atom can give an impression of the constraints that govern the buildup of the periodic table.the limits on the occupation of the subshells arise from the quantum numbers for the atomic electrons and their relationship to each other. The maximum number of electrons present in a particular shell is calculated by the formula 2n 2 , where "n" represents the shell number. The electronic configuration diagram represents an element in its ground state or stable state. Argon atom 22n chlorine atom chlorine ion 19 potassium atom potassium ion 3. Red, and white balls represent the c, n, fe, o, and h atoms, respectively... Rough sketches of the electron density for the first three shells of the hydrogen atom can give an impression of the constraints that govern the buildup of the periodic table.the limits on the occupation of the subshells arise from the quantum numbers for the atomic electrons and their relationship to each other.

Electronic configuration refers to the distribution of electrons in various shells/orbits/energy levels of every atom. Rough sketches of the electron density for the first three shells of the hydrogen atom can give an impression of the constraints that govern the buildup of the periodic table.the limits on the occupation of the subshells arise from the quantum numbers for the atomic electrons and their relationship to each other. Red, and white balls represent the c, n, fe, o, and h atoms, respectively. The electronic configuration diagram represents an element in its ground state or stable state. Atom/ion neon atom fluorine atom fluorine ion sodium atom sodium ion argon atom chlorine atom chlorine ion potassium atom potassium ion atomic number number of 10 protons 10 2.

Electronic configuration refers to the distribution of electrons in various shells/orbits/energy levels of every atom... Electronegativity is a measure of the tendency of an atom to attract a bonding pair of electrons. There is no chemical reaction but there ia a reaction in that the disassociation of the sodium and hydroxide ions and the hydration of those ions releases a lot of heat, enough to boil water in some circumstances. Use the table above to draw the bohr model diagram for the following atoms and ions. The electronic configuration diagram represents an element in its ground state or stable state. The pauling scale is the most commonly used. Argon atom 22n chlorine atom chlorine ion 19 potassium atom potassium ion 3. The maximum number of electrons present in a particular shell is calculated by the formula 2n 2 , where "n" represents the shell number... There are a set of rules to remember while distribution off electrons in different orbits.

Atom/ion neon atom fluorine atom fluorine ion sodium atom sodium ion argon atom chlorine atom chlorine ion potassium atom potassium ion atomic number number of 10 protons 10 2. Electronegativity is a measure of the tendency of an atom to attract a bonding pair of electrons. Atom of every element have different electronic configurations based on the atomic number of each element. The pauling scale is the most commonly used. Use the table above to draw the bohr model diagram for the following atoms and ions. Rough sketches of the electron density for the first three shells of the hydrogen atom can give an impression of the constraints that govern the buildup of the periodic table.the limits on the occupation of the subshells arise from the quantum numbers for the atomic electrons and their relationship to each other. The electrons from one atom are shown as dots, … These sketches arise from the hydrogen.

These sketches arise from the hydrogen. The maximum number of electrons present in a particular shell is calculated by the formula 2n 2 , where "n" represents the shell number. These sketches arise from the hydrogen. The electrons from one atom are shown as dots, … Rough sketches of the electron density for the first three shells of the hydrogen atom can give an impression of the constraints that govern the buildup of the periodic table.the limits on the occupation of the subshells arise from the quantum numbers for the atomic electrons and their relationship to each other. Atom/ion neon atom fluorine atom fluorine ion sodium atom sodium ion argon atom chlorine atom chlorine ion potassium atom potassium ion atomic number number of 10 protons 10 2. There are a set of rules to remember while distribution off electrons in different orbits. Electronic configuration refers to the distribution of electrons in various shells/orbits/energy levels of every atom. Use the table above to draw the bohr model diagram for the following atoms and ions.

There is no chemical reaction but there ia a reaction in that the disassociation of the sodium and hydroxide ions and the hydration of those ions releases a lot of heat, enough to boil water in some circumstances. . Fluorine (the most electronegative element) is assigned a value of 4.0, and values range down to caesium and francium which are the least electronegative at 0.7.

Argon atom 22n chlorine atom chlorine ion 19 potassium atom potassium ion 3.. These sketches arise from the hydrogen. Rough sketches of the electron density for the first three shells of the hydrogen atom can give an impression of the constraints that govern the buildup of the periodic table.the limits on the occupation of the subshells arise from the quantum numbers for the atomic electrons and their relationship to each other. Atom/ion neon atom fluorine atom fluorine ion sodium atom sodium ion argon atom chlorine atom chlorine ion potassium atom potassium ion atomic number number of 10 protons 10 2. Electronegativity is a measure of the tendency of an atom to attract a bonding pair of electrons. The maximum number of electrons present in a particular shell is calculated by the formula 2n 2 , where "n" represents the shell number.. Rough sketches of the electron density for the first three shells of the hydrogen atom can give an impression of the constraints that govern the buildup of the periodic table.the limits on the occupation of the subshells arise from the quantum numbers for the atomic electrons and their relationship to each other.

The electronic configuration diagram represents an element in its ground state or stable state.. Electronegativity is a measure of the tendency of an atom to attract a bonding pair of electrons. The electronic configuration diagram represents an element in its ground state or stable state. Electronic configuration refers to the distribution of electrons in various shells/orbits/energy levels of every atom. The maximum number of electrons present in a particular shell is calculated by the formula 2n 2 , where "n" represents the shell number... Atom/ion neon atom fluorine atom fluorine ion sodium atom sodium ion argon atom chlorine atom chlorine ion potassium atom potassium ion atomic number number of 10 protons 10 2.

Electronegativity is a measure of the tendency of an atom to attract a bonding pair of electrons. The electrons from one atom are shown as dots, … Fluorine (the most electronegative element) is assigned a value of 4.0, and values range down to caesium and francium which are the least electronegative at 0.7. Rough sketches of the electron density for the first three shells of the hydrogen atom can give an impression of the constraints that govern the buildup of the periodic table.the limits on the occupation of the subshells arise from the quantum numbers for the atomic electrons and their relationship to each other. The maximum number of electrons present in a particular shell is calculated by the formula 2n 2 , where "n" represents the shell number. Electronic configuration refers to the distribution of electrons in various shells/orbits/energy levels of every atom. Atom of every element have different electronic configurations based on the atomic number of each element. There is no chemical reaction but there ia a reaction in that the disassociation of the sodium and hydroxide ions and the hydration of those ions releases a lot of heat, enough to boil water in some circumstances. These sketches arise from the hydrogen. Electronegativity is a measure of the tendency of an atom to attract a bonding pair of electrons. There are a set of rules to remember while distribution off electrons in different orbits.

Electronic configuration refers to the distribution of electrons in various shells/orbits/energy levels of every atom. Use the table above to draw the bohr model diagram for the following atoms and ions. Atom of every element have different electronic configurations based on the atomic number of each element. There is no chemical reaction but there ia a reaction in that the disassociation of the sodium and hydroxide ions and the hydration of those ions releases a lot of heat, enough to boil water in some circumstances.

The electrons from one atom are shown as dots, … Atom of every element have different electronic configurations based on the atomic number of each element. Electronegativity is a measure of the tendency of an atom to attract a bonding pair of electrons. The pauling scale is the most commonly used. Use the table above to draw the bohr model diagram for the following atoms and ions. Electronic configuration refers to the distribution of electrons in various shells/orbits/energy levels of every atom. These sketches arise from the hydrogen. The electronic configuration diagram represents an element in its ground state or stable state... Atom of every element have different electronic configurations based on the atomic number of each element.

These sketches arise from the hydrogen. Electronegativity is a measure of the tendency of an atom to attract a bonding pair of electrons. There is no chemical reaction but there ia a reaction in that the disassociation of the sodium and hydroxide ions and the hydration of those ions releases a lot of heat, enough to boil water in some circumstances. Atom/ion neon atom fluorine atom fluorine ion sodium atom sodium ion argon atom chlorine atom chlorine ion potassium atom potassium ion atomic number number of 10 protons 10 2. Fluorine (the most electronegative element) is assigned a value of 4.0, and values range down to caesium and francium which are the least electronegative at 0.7.. Electronegativity is a measure of the tendency of an atom to attract a bonding pair of electrons.

The electronic configuration diagram represents an element in its ground state or stable state.. Fluorine (the most electronegative element) is assigned a value of 4.0, and values range down to caesium and francium which are the least electronegative at 0.7... There are a set of rules to remember while distribution off electrons in different orbits.

The maximum number of electrons present in a particular shell is calculated by the formula 2n 2 , where "n" represents the shell number. Atom of every element have different electronic configurations based on the atomic number of each element. The pauling scale is the most commonly used. The maximum number of electrons present in a particular shell is calculated by the formula 2n 2 , where "n" represents the shell number. Electronegativity is a measure of the tendency of an atom to attract a bonding pair of electrons. These sketches arise from the hydrogen. Argon atom 22n chlorine atom chlorine ion 19 potassium atom potassium ion 3. Rough sketches of the electron density for the first three shells of the hydrogen atom can give an impression of the constraints that govern the buildup of the periodic table.the limits on the occupation of the subshells arise from the quantum numbers for the atomic electrons and their relationship to each other. Use the table above to draw the bohr model diagram for the following atoms and ions... The electronic configuration diagram represents an element in its ground state or stable state.

Atom of every element have different electronic configurations based on the atomic number of each element. The electrons from one atom are shown as dots, … Use the table above to draw the bohr model diagram for the following atoms and ions. These sketches arise from the hydrogen. Electronic configuration refers to the distribution of electrons in various shells/orbits/energy levels of every atom... Rough sketches of the electron density for the first three shells of the hydrogen atom can give an impression of the constraints that govern the buildup of the periodic table.the limits on the occupation of the subshells arise from the quantum numbers for the atomic electrons and their relationship to each other.

Argon atom 22n chlorine atom chlorine ion 19 potassium atom potassium ion 3. Fluorine (the most electronegative element) is assigned a value of 4.0, and values range down to caesium and francium which are the least electronegative at 0.7... There is no chemical reaction but there ia a reaction in that the disassociation of the sodium and hydroxide ions and the hydration of those ions releases a lot of heat, enough to boil water in some circumstances.

The maximum number of electrons present in a particular shell is calculated by the formula 2n 2 , where "n" represents the shell number. .. The electrons from one atom are shown as dots, …

The pauling scale is the most commonly used. The maximum number of electrons present in a particular shell is calculated by the formula 2n 2 , where "n" represents the shell number. Atom/ion neon atom fluorine atom fluorine ion sodium atom sodium ion argon atom chlorine atom chlorine ion potassium atom potassium ion atomic number number of 10 protons 10 2. There is no chemical reaction but there ia a reaction in that the disassociation of the sodium and hydroxide ions and the hydration of those ions releases a lot of heat, enough to boil water in some circumstances. Rough sketches of the electron density for the first three shells of the hydrogen atom can give an impression of the constraints that govern the buildup of the periodic table.the limits on the occupation of the subshells arise from the quantum numbers for the atomic electrons and their relationship to each other.. The electrons from one atom are shown as dots, …

Argon atom 22n chlorine atom chlorine ion 19 potassium atom potassium ion 3.. Electronic configuration refers to the distribution of electrons in various shells/orbits/energy levels of every atom. These sketches arise from the hydrogen. Argon atom 22n chlorine atom chlorine ion 19 potassium atom potassium ion 3. The maximum number of electrons present in a particular shell is calculated by the formula 2n 2 , where "n" represents the shell number. Fluorine (the most electronegative element) is assigned a value of 4.0, and values range down to caesium and francium which are the least electronegative at 0.7. The electrons from one atom are shown as dots, …. The pauling scale is the most commonly used.

Red, and white balls represent the c, n, fe, o, and h atoms, respectively. These sketches arise from the hydrogen. Atom of every element have different electronic configurations based on the atomic number of each element. Rough sketches of the electron density for the first three shells of the hydrogen atom can give an impression of the constraints that govern the buildup of the periodic table.the limits on the occupation of the subshells arise from the quantum numbers for the atomic electrons and their relationship to each other.. The electrons from one atom are shown as dots, …

Atom of every element have different electronic configurations based on the atomic number of each element. The electronic configuration diagram represents an element in its ground state or stable state.. The electrons from one atom are shown as dots, …

Red, and white balls represent the c, n, fe, o, and h atoms, respectively.. Red, and white balls represent the c, n, fe, o, and h atoms, respectively. Atom/ion neon atom fluorine atom fluorine ion sodium atom sodium ion argon atom chlorine atom chlorine ion potassium atom potassium ion atomic number number of 10 protons 10 2. Electronegativity is a measure of the tendency of an atom to attract a bonding pair of electrons. The pauling scale is the most commonly used. The electronic configuration diagram represents an element in its ground state or stable state. Argon atom 22n chlorine atom chlorine ion 19 potassium atom potassium ion 3. Electronic configuration refers to the distribution of electrons in various shells/orbits/energy levels of every atom. Fluorine (the most electronegative element) is assigned a value of 4.0, and values range down to caesium and francium which are the least electronegative at 0.7. These sketches arise from the hydrogen. The electronic configuration diagram represents an element in its ground state or stable state.