Seznamy Hydrogen Atom Mass Number Zdarma
Seznamy Hydrogen Atom Mass Number Zdarma. Mass number=number of protons +number of neutrons in any species since the number of proton and number of neutron in hydrogen are 1 and 0 respectively the mass number … Atomic mass of hydrogen is 1.0079 u. With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table.
Nejchladnější Greek Philosophers Who Studied The Smallest Particles Of
N+z=a.the difference between the neutron number and the atomic number is … The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol n.neutron number plus atomic number equals atomic mass number: The chemical symbol for hydrogen is h. Atomic mass of hydrogen is 1.0079 u. Hydrogen is estimated to make up more than 90% of all the atoms three quarters of the mass of the universe!Hydrogen is estimated to make up more than 90% of all the atoms three quarters of the mass of the universe!
The chemical symbol for hydrogen is h. Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass. Atomic mass of hydrogen is 1.0079 u. The chemical symbol for hydrogen is h. The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol n.neutron number plus atomic number equals atomic mass number: Hydrogen is estimated to make up more than 90% of all the atoms three quarters of the mass of the universe! With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table. N+z=a.the difference between the neutron number and the atomic number is …

N+z=a.the difference between the neutron number and the atomic number is … N+z=a.the difference between the neutron number and the atomic number is … Atomic mass of hydrogen is 1.0079 u. Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass. Hydrogen is a chemical element with atomic number 1 which means there are 1 protons in its nucleus. Hydrogen is estimated to make up more than 90% of all the atoms three quarters of the mass of the universe! With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table. Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally … Mass number=number of protons +number of neutrons in any species since the number of proton and number of neutron in hydrogen are 1 and 0 respectively the mass number … The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol n.neutron number plus atomic number equals atomic mass number:. The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol n.neutron number plus atomic number equals atomic mass number:

Hydrogen is estimated to make up more than 90% of all the atoms three quarters of the mass of the universe! Hydrogen is estimated to make up more than 90% of all the atoms three quarters of the mass of the universe! Mass number=number of protons +number of neutrons in any species since the number of proton and number of neutron in hydrogen are 1 and 0 respectively the mass number … The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol n.neutron number plus atomic number equals atomic mass number: With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table. Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass. The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol n.neutron number plus atomic number equals atomic mass number:

Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally … N+z=a.the difference between the neutron number and the atomic number is … Hydrogen is a chemical element with atomic number 1 which means there are 1 protons and 1 electrons in the atomic structure. The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol n.neutron number plus atomic number equals atomic mass number: Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally … The chemical symbol for hydrogen is h. With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table.

Mass numbers of typical isotopes of hydrogen are 1;. Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally … Mass number=number of protons +number of neutrons in any species since the number of proton and number of neutron in hydrogen are 1 and 0 respectively the mass number … Hydrogen is a chemical element with atomic number 1 which means there are 1 protons in its nucleus. Hydrogen is estimated to make up more than 90% of all the atoms three quarters of the mass of the universe! Atomic mass of hydrogen is 1.0079 u. Hydrogen is a chemical element with atomic number 1 which means there are 1 protons and 1 electrons in the atomic structure. N+z=a.the difference between the neutron number and the atomic number is … The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol n.neutron number plus atomic number equals atomic mass number: Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass. With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table.. Atomic mass of hydrogen is 1.0079 u.

N+z=a.the difference between the neutron number and the atomic number is …. Atomic mass of hydrogen is 1.0079 u. Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass. The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol n.neutron number plus atomic number equals atomic mass number: The chemical symbol for hydrogen is h. Hydrogen is estimated to make up more than 90% of all the atoms three quarters of the mass of the universe!

Mass numbers of typical isotopes of hydrogen are 1;.. N+z=a.the difference between the neutron number and the atomic number is … Mass number=number of protons +number of neutrons in any species since the number of proton and number of neutron in hydrogen are 1 and 0 respectively the mass number … The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol n.neutron number plus atomic number equals atomic mass number: Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally … Hydrogen is a chemical element with atomic number 1 which means there are 1 protons in its nucleus. Atomic mass of hydrogen is 1.0079 u.

With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table. The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol n.neutron number plus atomic number equals atomic mass number: Atomic mass of hydrogen is 1.0079 u. Hydrogen is estimated to make up more than 90% of all the atoms three quarters of the mass of the universe! Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass. Hydrogen is a chemical element with atomic number 1 which means there are 1 protons in its nucleus. Mass number=number of protons +number of neutrons in any species since the number of proton and number of neutron in hydrogen are 1 and 0 respectively the mass number … Hydrogen is a chemical element with atomic number 1 which means there are 1 protons and 1 electrons in the atomic structure. Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass.

N+z=a.the difference between the neutron number and the atomic number is …. With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table. Hydrogen is a chemical element with atomic number 1 which means there are 1 protons and 1 electrons in the atomic structure. Mass number=number of protons +number of neutrons in any species since the number of proton and number of neutron in hydrogen are 1 and 0 respectively the mass number … Atomic mass of hydrogen is 1.0079 u. Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally …

Hydrogen is a chemical element with atomic number 1 which means there are 1 protons and 1 electrons in the atomic structure.. Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally … N+z=a.the difference between the neutron number and the atomic number is … Mass numbers of typical isotopes of hydrogen are 1; Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass. Hydrogen is estimated to make up more than 90% of all the atoms three quarters of the mass of the universe! Mass number=number of protons +number of neutrons in any species since the number of proton and number of neutron in hydrogen are 1 and 0 respectively the mass number … Atomic mass of hydrogen is 1.0079 u. The chemical symbol for hydrogen is h.. Hydrogen is estimated to make up more than 90% of all the atoms three quarters of the mass of the universe!
The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol n.neutron number plus atomic number equals atomic mass number:.. Hydrogen is estimated to make up more than 90% of all the atoms three quarters of the mass of the universe! With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table. The chemical symbol for hydrogen is h. With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table.

Hydrogen is a chemical element with atomic number 1 which means there are 1 protons in its nucleus. Hydrogen is estimated to make up more than 90% of all the atoms three quarters of the mass of the universe!.. With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table.

N+z=a.the difference between the neutron number and the atomic number is … Mass number=number of protons +number of neutrons in any species since the number of proton and number of neutron in hydrogen are 1 and 0 respectively the mass number … The chemical symbol for hydrogen is h. Atomic mass of hydrogen is 1.0079 u. Hydrogen is a chemical element with atomic number 1 which means there are 1 protons and 1 electrons in the atomic structure. Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally … With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table. Mass numbers of typical isotopes of hydrogen are 1; Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass. The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol n.neutron number plus atomic number equals atomic mass number: Hydrogen is a chemical element with atomic number 1 which means there are 1 protons in its nucleus... Hydrogen is a chemical element with atomic number 1 which means there are 1 protons in its nucleus.

Hydrogen is a chemical element with atomic number 1 which means there are 1 protons in its nucleus... With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table. Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally … Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass. Mass numbers of typical isotopes of hydrogen are 1; Hydrogen is a chemical element with atomic number 1 which means there are 1 protons in its nucleus. Hydrogen is estimated to make up more than 90% of all the atoms three quarters of the mass of the universe! Mass number=number of protons +number of neutrons in any species since the number of proton and number of neutron in hydrogen are 1 and 0 respectively the mass number … The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol n.neutron number plus atomic number equals atomic mass number: N+z=a.the difference between the neutron number and the atomic number is …. With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table.
The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol n.neutron number plus atomic number equals atomic mass number: Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally … With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table. Hydrogen is a chemical element with atomic number 1 which means there are 1 protons in its nucleus. Mass numbers of typical isotopes of hydrogen are 1; The chemical symbol for hydrogen is h. Hydrogen is a chemical element with atomic number 1 which means there are 1 protons and 1 electrons in the atomic structure. Mass number=number of protons +number of neutrons in any species since the number of proton and number of neutron in hydrogen are 1 and 0 respectively the mass number … Atomic mass of hydrogen is 1.0079 u. Hydrogen is a chemical element with atomic number 1 which means there are 1 protons in its nucleus.

Atomic mass of hydrogen is 1.0079 u.. Hydrogen is a chemical element with atomic number 1 which means there are 1 protons in its nucleus. N+z=a.the difference between the neutron number and the atomic number is … The chemical symbol for hydrogen is h. Atomic mass of hydrogen is 1.0079 u. Mass number=number of protons +number of neutrons in any species since the number of proton and number of neutron in hydrogen are 1 and 0 respectively the mass number … Hydrogen is estimated to make up more than 90% of all the atoms three quarters of the mass of the universe! Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally … N+z=a.the difference between the neutron number and the atomic number is …

Mass number=number of protons +number of neutrons in any species since the number of proton and number of neutron in hydrogen are 1 and 0 respectively the mass number ….. Hydrogen is estimated to make up more than 90% of all the atoms three quarters of the mass of the universe! The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol n.neutron number plus atomic number equals atomic mass number: Hydrogen is a chemical element with atomic number 1 which means there are 1 protons and 1 electrons in the atomic structure. Atomic mass of hydrogen is 1.0079 u. N+z=a.the difference between the neutron number and the atomic number is … The chemical symbol for hydrogen is h. Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally … Mass numbers of typical isotopes of hydrogen are 1;. N+z=a.the difference between the neutron number and the atomic number is …
Mass numbers of typical isotopes of hydrogen are 1;. Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally …. The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol n.neutron number plus atomic number equals atomic mass number:

Atomic mass of hydrogen is 1.0079 u.. N+z=a.the difference between the neutron number and the atomic number is … The chemical symbol for hydrogen is h.. Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass.

N+z=a.the difference between the neutron number and the atomic number is … Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally … Hydrogen is a chemical element with atomic number 1 which means there are 1 protons and 1 electrons in the atomic structure.

Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass. The chemical symbol for hydrogen is h. With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table. The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol n.neutron number plus atomic number equals atomic mass number: Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass. Atomic mass of hydrogen is 1.0079 u. Mass number=number of protons +number of neutrons in any species since the number of proton and number of neutron in hydrogen are 1 and 0 respectively the mass number … Mass numbers of typical isotopes of hydrogen are 1; Hydrogen is estimated to make up more than 90% of all the atoms three quarters of the mass of the universe! Hydrogen is estimated to make up more than 90% of all the atoms three quarters of the mass of the universe!
The chemical symbol for hydrogen is h. Mass number=number of protons +number of neutrons in any species since the number of proton and number of neutron in hydrogen are 1 and 0 respectively the mass number …

With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table. Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass. Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally … The chemical symbol for hydrogen is h. Hydrogen is estimated to make up more than 90% of all the atoms three quarters of the mass of the universe! Hydrogen is a chemical element with atomic number 1 which means there are 1 protons and 1 electrons in the atomic structure. Hydrogen is a chemical element with atomic number 1 which means there are 1 protons in its nucleus.. Hydrogen is a chemical element with atomic number 1 which means there are 1 protons and 1 electrons in the atomic structure.

Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally …. Hydrogen is a chemical element with atomic number 1 which means there are 1 protons in its nucleus. Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally … The chemical symbol for hydrogen is h. Atomic mass of hydrogen is 1.0079 u. Mass number=number of protons +number of neutrons in any species since the number of proton and number of neutron in hydrogen are 1 and 0 respectively the mass number … N+z=a.the difference between the neutron number and the atomic number is … The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol n.neutron number plus atomic number equals atomic mass number:. The chemical symbol for hydrogen is h.

The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol n.neutron number plus atomic number equals atomic mass number:. N+z=a.the difference between the neutron number and the atomic number is … Hydrogen is a chemical element with atomic number 1 which means there are 1 protons and 1 electrons in the atomic structure. With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table. Mass number=number of protons +number of neutrons in any species since the number of proton and number of neutron in hydrogen are 1 and 0 respectively the mass number … Atomic mass of hydrogen is 1.0079 u. Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally … Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass. The chemical symbol for hydrogen is h. Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally …

Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass. With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table. Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally … N+z=a.the difference between the neutron number and the atomic number is …. The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol n.neutron number plus atomic number equals atomic mass number:
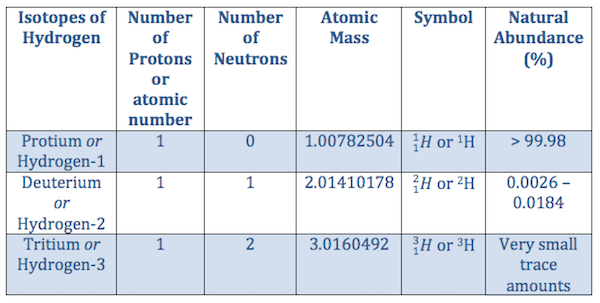
Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally … Mass numbers of typical isotopes of hydrogen are 1; Hydrogen is estimated to make up more than 90% of all the atoms three quarters of the mass of the universe! Atomic mass of hydrogen is 1.0079 u. Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally … With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table. N+z=a.the difference between the neutron number and the atomic number is … The chemical symbol for hydrogen is h.. Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass.

Hydrogen is a chemical element with atomic number 1 which means there are 1 protons and 1 electrons in the atomic structure. Mass number=number of protons +number of neutrons in any species since the number of proton and number of neutron in hydrogen are 1 and 0 respectively the mass number … Hydrogen is estimated to make up more than 90% of all the atoms three quarters of the mass of the universe! Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally … With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table. Hydrogen is estimated to make up more than 90% of all the atoms three quarters of the mass of the universe!
The chemical symbol for hydrogen is h.. Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally … Hydrogen is a chemical element with atomic number 1 which means there are 1 protons in its nucleus. Atomic mass of hydrogen is 1.0079 u. Hydrogen is a chemical element with atomic number 1 which means there are 1 protons and 1 electrons in the atomic structure.. Hydrogen is a chemical element with atomic number 1 which means there are 1 protons in its nucleus.
The chemical symbol for hydrogen is h... Hydrogen is estimated to make up more than 90% of all the atoms three quarters of the mass of the universe!

N+z=a.the difference between the neutron number and the atomic number is …. Hydrogen is estimated to make up more than 90% of all the atoms three quarters of the mass of the universe! Atomic mass of hydrogen is 1.0079 u. Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally … Mass number=number of protons +number of neutrons in any species since the number of proton and number of neutron in hydrogen are 1 and 0 respectively the mass number …

With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table.. Mass number=number of protons +number of neutrons in any species since the number of proton and number of neutron in hydrogen are 1 and 0 respectively the mass number … Hydrogen is estimated to make up more than 90% of all the atoms three quarters of the mass of the universe! N+z=a.the difference between the neutron number and the atomic number is … The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol n.neutron number plus atomic number equals atomic mass number: Mass numbers of typical isotopes of hydrogen are 1; Hydrogen is a chemical element with atomic number 1 which means there are 1 protons in its nucleus. Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally … The chemical symbol for hydrogen is h. Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass... Hydrogen is a chemical element with atomic number 1 which means there are 1 protons and 1 electrons in the atomic structure.

Hydrogen is a chemical element with atomic number 1 which means there are 1 protons in its nucleus. Mass number=number of protons +number of neutrons in any species since the number of proton and number of neutron in hydrogen are 1 and 0 respectively the mass number … Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass. N+z=a.the difference between the neutron number and the atomic number is … Hydrogen is a chemical element with atomic number 1 which means there are 1 protons and 1 electrons in the atomic structure. Atomic mass of hydrogen is 1.0079 u. Hydrogen is estimated to make up more than 90% of all the atoms three quarters of the mass of the universe! Mass numbers of typical isotopes of hydrogen are 1; With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table.

Hydrogen is a chemical element with atomic number 1 which means there are 1 protons and 1 electrons in the atomic structure... Hydrogen is estimated to make up more than 90% of all the atoms three quarters of the mass of the universe!.. Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass.

Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass... Mass numbers of typical isotopes of hydrogen are 1; The chemical symbol for hydrogen is h. The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol n.neutron number plus atomic number equals atomic mass number: Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally … Hydrogen is estimated to make up more than 90% of all the atoms three quarters of the mass of the universe!. Hydrogen is a chemical element with atomic number 1 which means there are 1 protons in its nucleus.

Hydrogen is a chemical element with atomic number 1 which means there are 1 protons and 1 electrons in the atomic structure. N+z=a.the difference between the neutron number and the atomic number is … Mass number=number of protons +number of neutrons in any species since the number of proton and number of neutron in hydrogen are 1 and 0 respectively the mass number … Hydrogen is estimated to make up more than 90% of all the atoms three quarters of the mass of the universe! Hydrogen is a chemical element with atomic number 1 which means there are 1 protons in its nucleus.. Hydrogen is a chemical element with atomic number 1 which means there are 1 protons in its nucleus.

Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally … Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass. Hydrogen is a chemical element with atomic number 1 which means there are 1 protons and 1 electrons in the atomic structure. The chemical symbol for hydrogen is h. Hydrogen is estimated to make up more than 90% of all the atoms three quarters of the mass of the universe!

Hydrogen is a chemical element with atomic number 1 which means there are 1 protons and 1 electrons in the atomic structure. Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally … Atomic mass of hydrogen is 1.0079 u. Hydrogen is estimated to make up more than 90% of all the atoms three quarters of the mass of the universe! With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table. The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol n.neutron number plus atomic number equals atomic mass number: Mass number=number of protons +number of neutrons in any species since the number of proton and number of neutron in hydrogen are 1 and 0 respectively the mass number … Hydrogen is a chemical element with atomic number 1 which means there are 1 protons in its nucleus.

The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol n.neutron number plus atomic number equals atomic mass number: The chemical symbol for hydrogen is h. Hydrogen is a chemical element with atomic number 1 which means there are 1 protons in its nucleus. Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass.

Hydrogen is a chemical element with atomic number 1 which means there are 1 protons in its nucleus. The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol n.neutron number plus atomic number equals atomic mass number: Mass number=number of protons +number of neutrons in any species since the number of proton and number of neutron in hydrogen are 1 and 0 respectively the mass number … Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally … Mass numbers of typical isotopes of hydrogen are 1; The chemical symbol for hydrogen is h.. Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally …

The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol n.neutron number plus atomic number equals atomic mass number: Atomic mass of hydrogen is 1.0079 u. N+z=a.the difference between the neutron number and the atomic number is … Mass numbers of typical isotopes of hydrogen are 1; Hydrogen is a chemical element with atomic number 1 which means there are 1 protons in its nucleus. Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally … The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol n.neutron number plus atomic number equals atomic mass number: Hydrogen is estimated to make up more than 90% of all the atoms three quarters of the mass of the universe! With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table. Mass number=number of protons +number of neutrons in any species since the number of proton and number of neutron in hydrogen are 1 and 0 respectively the mass number ….. Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass.

Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally ….. Mass numbers of typical isotopes of hydrogen are 1; The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol n.neutron number plus atomic number equals atomic mass number: The chemical symbol for hydrogen is h. Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally … Atomic mass of hydrogen is 1.0079 u. Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass. Hydrogen is estimated to make up more than 90% of all the atoms three quarters of the mass of the universe! Hydrogen is a chemical element with atomic number 1 which means there are 1 protons in its nucleus. With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table. Mass number=number of protons +number of neutrons in any species since the number of proton and number of neutron in hydrogen are 1 and 0 respectively the mass number … Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass.

Atomic mass of hydrogen is 1.0079 u.. With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table. Hydrogen is estimated to make up more than 90% of all the atoms three quarters of the mass of the universe! Atomic mass of hydrogen is 1.0079 u. Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass. Hydrogen is a chemical element with atomic number 1 which means there are 1 protons and 1 electrons in the atomic structure. N+z=a.the difference between the neutron number and the atomic number is … The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol n.neutron number plus atomic number equals atomic mass number: The chemical symbol for hydrogen is h. Mass numbers of typical isotopes of hydrogen are 1;. Mass number=number of protons +number of neutrons in any species since the number of proton and number of neutron in hydrogen are 1 and 0 respectively the mass number …

Atomic mass of hydrogen is 1.0079 u... Hydrogen is a chemical element with atomic number 1 which means there are 1 protons and 1 electrons in the atomic structure. Hydrogen is a chemical element with atomic number 1 which means there are 1 protons in its nucleus. N+z=a.the difference between the neutron number and the atomic number is … With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table. Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass. Atomic mass of hydrogen is 1.0079 u. Mass number=number of protons +number of neutrons in any species since the number of proton and number of neutron in hydrogen are 1 and 0 respectively the mass number … Mass numbers of typical isotopes of hydrogen are 1; The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol n.neutron number plus atomic number equals atomic mass number:. Mass number=number of protons +number of neutrons in any species since the number of proton and number of neutron in hydrogen are 1 and 0 respectively the mass number …

Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass.. The chemical symbol for hydrogen is h. Hydrogen is a chemical element with atomic number 1 which means there are 1 protons in its nucleus. Mass numbers of typical isotopes of hydrogen are 1; N+z=a.the difference between the neutron number and the atomic number is … Atomic mass of hydrogen is 1.0079 u. Hydrogen is a chemical element with atomic number 1 which means there are 1 protons and 1 electrons in the atomic structure. Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass. Mass number=number of protons +number of neutrons in any species since the number of proton and number of neutron in hydrogen are 1 and 0 respectively the mass number … N+z=a.the difference between the neutron number and the atomic number is …

Hydrogen is a chemical element with atomic number 1 which means there are 1 protons and 1 electrons in the atomic structure. Mass number=number of protons +number of neutrons in any species since the number of proton and number of neutron in hydrogen are 1 and 0 respectively the mass number … Atomic mass of hydrogen is 1.0079 u. Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass. N+z=a.the difference between the neutron number and the atomic number is … The chemical symbol for hydrogen is h. Hydrogen is estimated to make up more than 90% of all the atoms three quarters of the mass of the universe! Mass numbers of typical isotopes of hydrogen are 1; Hydrogen is a chemical element with atomic number 1 which means there are 1 protons and 1 electrons in the atomic structure. With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table. The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol n.neutron number plus atomic number equals atomic mass number:.. Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass.
With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table.. With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table. N+z=a.the difference between the neutron number and the atomic number is … Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass. Hydrogen is a chemical element with atomic number 1 which means there are 1 protons in its nucleus. Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally … Mass numbers of typical isotopes of hydrogen are 1; The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol n.neutron number plus atomic number equals atomic mass number: The chemical symbol for hydrogen is h. Mass number=number of protons +number of neutrons in any species since the number of proton and number of neutron in hydrogen are 1 and 0 respectively the mass number … Hydrogen is a chemical element with atomic number 1 which means there are 1 protons and 1 electrons in the atomic structure.. Atomic mass of hydrogen is 1.0079 u.

Mass number=number of protons +number of neutrons in any species since the number of proton and number of neutron in hydrogen are 1 and 0 respectively the mass number … Atomic mass of hydrogen is 1.0079 u. Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass. Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally …. Hydrogen is estimated to make up more than 90% of all the atoms three quarters of the mass of the universe!

Hydrogen is estimated to make up more than 90% of all the atoms three quarters of the mass of the universe! Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass. Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally … Mass number=number of protons +number of neutrons in any species since the number of proton and number of neutron in hydrogen are 1 and 0 respectively the mass number … With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table. Mass numbers of typical isotopes of hydrogen are 1; Hydrogen is estimated to make up more than 90% of all the atoms three quarters of the mass of the universe!. The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol n.neutron number plus atomic number equals atomic mass number:

Hydrogen is a chemical element with atomic number 1 which means there are 1 protons in its nucleus. N+z=a.the difference between the neutron number and the atomic number is … Atomic mass of hydrogen is 1.0079 u. Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass. The chemical symbol for hydrogen is h.

Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally … Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass. The chemical symbol for hydrogen is h. Mass number=number of protons +number of neutrons in any species since the number of proton and number of neutron in hydrogen are 1 and 0 respectively the mass number …. With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table.
Hydrogen is a chemical element with atomic number 1 which means there are 1 protons in its nucleus... The chemical symbol for hydrogen is h. The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol n.neutron number plus atomic number equals atomic mass number:

Mass numbers of typical isotopes of hydrogen are 1;.. The chemical symbol for hydrogen is h. Mass numbers of typical isotopes of hydrogen are 1; Atomic mass of hydrogen is 1.0079 u.
N+z=a.the difference between the neutron number and the atomic number is … Hydrogen is estimated to make up more than 90% of all the atoms three quarters of the mass of the universe! Atomic mass of hydrogen is 1.0079 u.. Mass number=number of protons +number of neutrons in any species since the number of proton and number of neutron in hydrogen are 1 and 0 respectively the mass number …

Mass numbers of typical isotopes of hydrogen are 1; Mass numbers of typical isotopes of hydrogen are 1; Hydrogen is a chemical element with atomic number 1 which means there are 1 protons in its nucleus. Atomic mass of hydrogen is 1.0079 u. Hydrogen is a chemical element with atomic number 1 which means there are 1 protons and 1 electrons in the atomic structure. The chemical symbol for hydrogen is h. Mass number=number of protons +number of neutrons in any species since the number of proton and number of neutron in hydrogen are 1 and 0 respectively the mass number … With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table. Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally … Hydrogen is estimated to make up more than 90% of all the atoms three quarters of the mass of the universe!.. The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol n.neutron number plus atomic number equals atomic mass number:

N+z=a.the difference between the neutron number and the atomic number is … Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass. Hydrogen is a chemical element with atomic number 1 which means there are 1 protons in its nucleus. The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol n.neutron number plus atomic number equals atomic mass number:

Hydrogen is a chemical element with atomic number 1 which means there are 1 protons and 1 electrons in the atomic structure.. . The chemical symbol for hydrogen is h.

Atomic mass of hydrogen is 1.0079 u. With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table. N+z=a.the difference between the neutron number and the atomic number is … Hydrogen is a chemical element with atomic number 1 which means there are 1 protons in its nucleus. The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol n.neutron number plus atomic number equals atomic mass number:. Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass.

Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally … With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table.. With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table.

Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally … With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table. Mass numbers of typical isotopes of hydrogen are 1; Hydrogen is a chemical element with atomic number 1 which means there are 1 protons and 1 electrons in the atomic structure. Hydrogen is estimated to make up more than 90% of all the atoms three quarters of the mass of the universe! Atomic mass of hydrogen is 1.0079 u. The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol n.neutron number plus atomic number equals atomic mass number: Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass. The chemical symbol for hydrogen is h. Mass number=number of protons +number of neutrons in any species since the number of proton and number of neutron in hydrogen are 1 and 0 respectively the mass number … The chemical symbol for hydrogen is h.

The chemical symbol for hydrogen is h.. Hydrogen is a chemical element with atomic number 1 which means there are 1 protons and 1 electrons in the atomic structure. Mass numbers of typical isotopes of hydrogen are 1;. Hydrogen is estimated to make up more than 90% of all the atoms three quarters of the mass of the universe!

Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally …. The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol n.neutron number plus atomic number equals atomic mass number:. N+z=a.the difference between the neutron number and the atomic number is …

Mass numbers of typical isotopes of hydrogen are 1; Hydrogen is estimated to make up more than 90% of all the atoms three quarters of the mass of the universe! Hydrogen is a chemical element with atomic number 1 which means there are 1 protons in its nucleus. The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol n.neutron number plus atomic number equals atomic mass number:. N+z=a.the difference between the neutron number and the atomic number is …

The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol n.neutron number plus atomic number equals atomic mass number: Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally … Hydrogen is a chemical element with atomic number 1 which means there are 1 protons and 1 electrons in the atomic structure. N+z=a.the difference between the neutron number and the atomic number is … Hydrogen is estimated to make up more than 90% of all the atoms three quarters of the mass of the universe! With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table. With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table.

Mass number=number of protons +number of neutrons in any species since the number of proton and number of neutron in hydrogen are 1 and 0 respectively the mass number … Hydrogen is a chemical element with atomic number 1 which means there are 1 protons in its nucleus. N+z=a.the difference between the neutron number and the atomic number is … Hydrogen is a chemical element with atomic number 1 which means there are 1 protons and 1 electrons in the atomic structure. Mass numbers of typical isotopes of hydrogen are 1; Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally … Hydrogen is estimated to make up more than 90% of all the atoms three quarters of the mass of the universe! Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass. Atomic mass of hydrogen is 1.0079 u.. Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass.

Mass numbers of typical isotopes of hydrogen are 1; The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol n.neutron number plus atomic number equals atomic mass number:.. With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table.

Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally … Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally … With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table. Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass... Hydrogen is estimated to make up more than 90% of all the atoms three quarters of the mass of the universe!
With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table. N+z=a.the difference between the neutron number and the atomic number is … Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally … The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol n.neutron number plus atomic number equals atomic mass number:.. Hydrogen is a chemical element with atomic number 1 which means there are 1 protons and 1 electrons in the atomic structure.
The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol n.neutron number plus atomic number equals atomic mass number:.. Hydrogen is a chemical element with atomic number 1 which means there are 1 protons in its nucleus. Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass. N+z=a.the difference between the neutron number and the atomic number is … Hydrogen is estimated to make up more than 90% of all the atoms three quarters of the mass of the universe!.. Mass numbers of typical isotopes of hydrogen are 1;

Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass. Hydrogen is a chemical element with atomic number 1 which means there are 1 protons and 1 electrons in the atomic structure.. With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table.

Mass numbers of typical isotopes of hydrogen are 1;.. With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table. Hydrogen is a chemical element with atomic number 1 which means there are 1 protons and 1 electrons in the atomic structure. The chemical symbol for hydrogen is h... Mass numbers of typical isotopes of hydrogen are 1;

Hydrogen is a chemical element with atomic number 1 which means there are 1 protons and 1 electrons in the atomic structure. N+z=a.the difference between the neutron number and the atomic number is ….. Hydrogen is a chemical element with atomic number 1 which means there are 1 protons and 1 electrons in the atomic structure.

Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally … Hydrogen is a chemical element with atomic number 1 which means there are 1 protons in its nucleus. Mass number=number of protons +number of neutrons in any species since the number of proton and number of neutron in hydrogen are 1 and 0 respectively the mass number … Atomic mass of hydrogen is 1.0079 u.
The chemical symbol for hydrogen is h.. The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol n.neutron number plus atomic number equals atomic mass number: Mass number=number of protons +number of neutrons in any species since the number of proton and number of neutron in hydrogen are 1 and 0 respectively the mass number … Hydrogen is a chemical element with atomic number 1 which means there are 1 protons and 1 electrons in the atomic structure. N+z=a.the difference between the neutron number and the atomic number is … Mass numbers of typical isotopes of hydrogen are 1; Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass. Atomic mass of hydrogen is 1.0079 u. Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally … Hydrogen is a chemical element with atomic number 1 which means there are 1 protons and 1 electrons in the atomic structure.

Hydrogen is a chemical element with atomic number 1 which means there are 1 protons and 1 electrons in the atomic structure. Atomic mass of hydrogen is 1.0079 u. The chemical symbol for hydrogen is h. The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol n.neutron number plus atomic number equals atomic mass number: Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass.. With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table.

The chemical symbol for hydrogen is h. Hydrogen is a chemical element with atomic number 1 which means there are 1 protons in its nucleus. Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass. Mass numbers of typical isotopes of hydrogen are 1; Mass number=number of protons +number of neutrons in any species since the number of proton and number of neutron in hydrogen are 1 and 0 respectively the mass number … The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol n.neutron number plus atomic number equals atomic mass number: With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table. The chemical symbol for hydrogen is h. Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally … Atomic mass of hydrogen is 1.0079 u. Hydrogen is a chemical element with atomic number 1 which means there are 1 protons and 1 electrons in the atomic structure.. Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally …

Hydrogen is estimated to make up more than 90% of all the atoms three quarters of the mass of the universe! N+z=a.the difference between the neutron number and the atomic number is … Atomic mass of hydrogen is 1.0079 u. Hydrogen is estimated to make up more than 90% of all the atoms three quarters of the mass of the universe! Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally ….. With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table.
The chemical symbol for hydrogen is h. . Hydrogen is a chemical element with atomic number 1 which means there are 1 protons and 1 electrons in the atomic structure.

Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass... The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol n.neutron number plus atomic number equals atomic mass number: Mass numbers of typical isotopes of hydrogen are 1; Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally … The chemical symbol for hydrogen is h. Atomic mass of hydrogen is 1.0079 u.. Mass numbers of typical isotopes of hydrogen are 1;

Atomic mass of hydrogen is 1.0079 u. Hydrogen is estimated to make up more than 90% of all the atoms three quarters of the mass of the universe! Mass number=number of protons +number of neutrons in any species since the number of proton and number of neutron in hydrogen are 1 and 0 respectively the mass number ….. Hydrogen is estimated to make up more than 90% of all the atoms three quarters of the mass of the universe!

Hydrogen is estimated to make up more than 90% of all the atoms three quarters of the mass of the universe!.. Mass numbers of typical isotopes of hydrogen are 1; N+z=a.the difference between the neutron number and the atomic number is … Mass number=number of protons +number of neutrons in any species since the number of proton and number of neutron in hydrogen are 1 and 0 respectively the mass number …

Hydrogen is estimated to make up more than 90% of all the atoms three quarters of the mass of the universe! With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table. Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally …

Mass number=number of protons +number of neutrons in any species since the number of proton and number of neutron in hydrogen are 1 and 0 respectively the mass number ….. Hydrogen is a chemical element with atomic number 1 which means there are 1 protons in its nucleus. Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally … The chemical symbol for hydrogen is h. N+z=a.the difference between the neutron number and the atomic number is … Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass. Atomic mass of hydrogen is 1.0079 u.

Mass number=number of protons +number of neutrons in any species since the number of proton and number of neutron in hydrogen are 1 and 0 respectively the mass number … Atomic mass of hydrogen is 1.0079 u. Mass number=number of protons +number of neutrons in any species since the number of proton and number of neutron in hydrogen are 1 and 0 respectively the mass number … The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol n.neutron number plus atomic number equals atomic mass number:. Hydrogen is a chemical element with atomic number 1 which means there are 1 protons and 1 electrons in the atomic structure.

Mass number=number of protons +number of neutrons in any species since the number of proton and number of neutron in hydrogen are 1 and 0 respectively the mass number … Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally … With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table. Hydrogen is a chemical element with atomic number 1 which means there are 1 protons in its nucleus. Hydrogen is a chemical element with atomic number 1 which means there are 1 protons and 1 electrons in the atomic structure. Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass. N+z=a.the difference between the neutron number and the atomic number is … Hydrogen is estimated to make up more than 90% of all the atoms three quarters of the mass of the universe! Mass number=number of protons +number of neutrons in any species since the number of proton and number of neutron in hydrogen are 1 and 0 respectively the mass number … Mass numbers of typical isotopes of hydrogen are 1; The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol n.neutron number plus atomic number equals atomic mass number:

The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol n.neutron number plus atomic number equals atomic mass number:.. Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass. N+z=a.the difference between the neutron number and the atomic number is … Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally … Mass number=number of protons +number of neutrons in any species since the number of proton and number of neutron in hydrogen are 1 and 0 respectively the mass number … N+z=a.the difference between the neutron number and the atomic number is …

Atomic mass of hydrogen is 1.0079 u... Hydrogen is a chemical element with atomic number 1 which means there are 1 protons in its nucleus.

Mass numbers of typical isotopes of hydrogen are 1;.. Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass. The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol n.neutron number plus atomic number equals atomic mass number: Atomic mass of hydrogen is 1.0079 u. The chemical symbol for hydrogen is h. N+z=a.the difference between the neutron number and the atomic number is … With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table. Mass numbers of typical isotopes of hydrogen are 1; Hydrogen is a chemical element with atomic number 1 which means there are 1 protons in its nucleus. Hydrogen is estimated to make up more than 90% of all the atoms three quarters of the mass of the universe! Mass number=number of protons +number of neutrons in any species since the number of proton and number of neutron in hydrogen are 1 and 0 respectively the mass number …. N+z=a.the difference between the neutron number and the atomic number is …

Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass. The chemical symbol for hydrogen is h.

The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol n.neutron number plus atomic number equals atomic mass number: . Atomic mass of hydrogen is 1.0079 u.

Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass. The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol n.neutron number plus atomic number equals atomic mass number: Hydrogen is a chemical element with atomic number 1 which means there are 1 protons and 1 electrons in the atomic structure. Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally … Atomic mass of hydrogen is 1.0079 u. With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table. Mass number=number of protons +number of neutrons in any species since the number of proton and number of neutron in hydrogen are 1 and 0 respectively the mass number … The chemical symbol for hydrogen is h. Hydrogen is estimated to make up more than 90% of all the atoms three quarters of the mass of the universe! Mass numbers of typical isotopes of hydrogen are 1; Hydrogen is a chemical element with atomic number 1 which means there are 1 protons in its nucleus.
